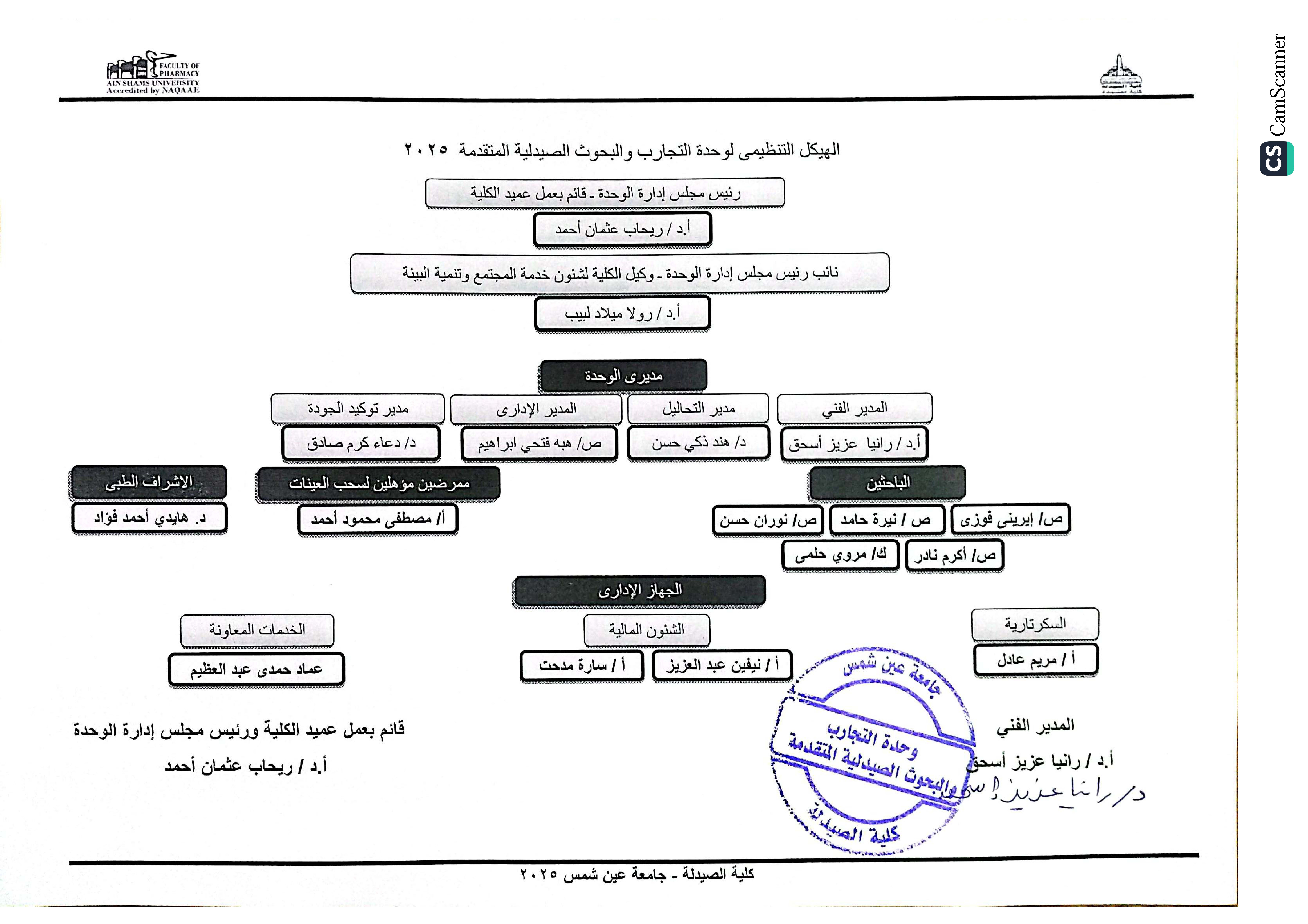
EAPRU License:-

The Advanced Pharmaceutical Experiments and Research Unit was established at the Faculty of Pharmacy, Ain Shams University, with the approval of the Supreme Council of Universities on October 25, 1997 as a unit of a special nature. The license was renewed by the Egyptian Ministry of Health on January 1, 2017.
The Advanced Pharmaceutical Experiments and Research Unit is an accredited research unit specialized in the field of pharmaceutical sciences. It is equipped with the latest technological devices. It also has a team consisting of experts in clinical analysis and follow-up who hold the highest scientific degrees such as doctorates, masters and diplomas in clinical pharmacy. The unit provided its services over the previous years to many pharmaceutical companies in the Arab Republic of Egypt as well as some Arab countries.

EAPRU Vision:-
To be one of the best international centers in the field of pharmaceutical studies.
EAPRU Mission:-
Providing the best analytical methods while ensuring that the necessary requirements of the regulatory guidelines are followed and ensuring that accurate and reliable results are reached.

The strategic goals of the Advanced Pharmaceutical Experiments and Research Unit (EAPRU):-
1- Assisting the university and the faculty in carrying out its mission in the field of student education and training, and in the field of pharmaceutical research and experiments.
2- Assisting and providing services to the community and the surrounding environment in terms of health care.
3- Conducting experiments and scientific research aimed at solving realistic problems faced by productive activity in various work sites related to pharmacy and health care.
4- Assisting productive activity with scientific methods and research that lead to the development and creation of new methods that lead to an abundance of production and an improvement in its quality.
5- Training of community members, especially pharmacists or health care personnel, to use innovative scientific and technical methods, research, and experiments, educate them, and raise their productivity in various fields.
6- Strengthening cultural and scientific ties with other universities and scientific bodies at the Egyptian, Arab and international levels.
7- Contribute to the implementation of university projects, faculties and institutes, and carry out the necessary research and experiments that fall within the unit's competence.
8- Carrying out experiments and research or service work for others in the fields of pharmacy and health care.
9- Carrying out experiments and research work in all fields assigned to it within the limits of its competence

Mechanisms to achieve the ethics of scientific research:-
1- Formalization of the members of Board of the Scientific Research Ethics Committee headed by Prof. Dr. / President of the University.
2- Presenting the protocols of the volunteers participating in the unit's research to the Scientific Research Ethics Committee before conducting the study.
3- Not to conduct studies on volunteers without prior approval from the Scientific Research Ethics Committee.
4- Follow up the study procedures to verify compliance with the ethics of scientific research.
5- Raising awareness and announcing the moral code and disseminating it to all the members working in the unit.
Mechanisms to regulate the rights and duties of volunteers in scientific research:-
1- Ensuring, through volunteer protocols, that all their civil, human and religious rights are available.
2- Follow up on the implementation of all steps and procedures with volunteer protocols to ensure their safety and rights.
3- Ensuring that volunteers are not harmed through medical supervision on the doses of medications given to them, as well as the side effects, whether in the short or long term.
4- Ensuring the realization of all women's rights stipulated in the Egyptian Law, by providing a female member in the Ethics Committee.
Mechanisms for accountability in cases of breach of the rights and duties of volunteers in scientific research:-
1- In the event of any disagreement between the volunteers, the Scientific Research Ethics Committee is entrusted with adjudicating these disputes, provided that it submits a detailed report to the Professor/Dean of the faculty. This is for presentation to the unit's Board of Directors.
2- The professor/dean of the faculty discusses the issue to take all corrective measures in light of the aforementioned rights and duties.


The role of the Advanced Pharmaceutical Experiments and Research Unit to serve the community:-
* Formation of a house of expertise through providing consultations to drug companies in the fields of pharmacy, drug industry, drug control, and drug registration requirements locally and globally.
* Developing research and various studies on the side effects of medicines, whether extracted from herbs and medicinal plants or chemical medicines.
* The participation of Ain Shams University and the Faculty of Pharmacy in carrying out its mission in the field of educating and training students in the field of research and providing services to the community.
* Contribute to the implementation of university and faculty projects by providing technical consultations in the pharmaceutical fields.
* Coordination between the unit and the various scientific departments that conduct research on medicines in order to reach the optimal use or development of these medicines.
* Confirming the quality of pharmaceutical preparations circulating in the local market, which will have an economic return on treatment costs.
* Conducting scientific research aimed at solving realistic problems faced by productive activity in various work sites in the community, which are related to the fields of pharmacy, health care and environmental protection, and contributing to combating toxins and environmental pollution as a community service and contribution to sustainable environmental development.
* Contribute to spreading the culture of optimal use of medicine.
Consultations carried out by the Advanced Pharmaceutical Experiments and Research Unit within a year:-
2009
|
Company Name |
Trade Name of Product |
No. |
|
MEPACO Company(B.E.) |
Vedocard® 3.125mg tablets |
1 |
|
Amriya Pharm Ind. Company(B.E.) |
Amristatin® 80mg tablets |
2 |
|
October pharma Company(B.E.) |
Glucoless® 4 mg tablets |
3 |
|
Glaxosmithkline Company(B.E.) |
Ibidroxil® 500mg/5ml Suspension |
4 |
|
Sedico pharmaceutical Company (B.E.) |
Magna-Biotic Plus ® 1gm tablets |
5 |
|
Amriya Pharm Ind. Company (B.E.) |
Microserc® 24mg capsules |
6 |
|
Egyptian group for pharmaceutical Industries Company (B.E.) |
Cefathird® 300mg capsules |
7 |
|
Egyptian group for pharmaceutical Industries Company (B.E.) |
Cefathird® 125mg/5ml suspension |
8 |
|
Sanofi Aventis Company (B.E.) |
Novaldol® 1000mg tablets |
9 |
|
ADWIA Company |
Right - ACE® tablets |
10 |
|
October Pharma Company (B.E.) |
Statirose® 20mg tablets |
11 |
|
October Pharma Company (B.E.) |
Sarcozide® tablets |
12 |
|
European Egyptian Pharm. Ind. Company (B.E.) |
Simvastatin® 80mg tablets |
13 |
|
MEDIPHAR LABORATORIES Company (B.E.) |
TALUSIN®0.4mg capsules |
14 |
2010
|
Company Name |
Trade Name of Product |
No. |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
APEMIN 15mg cap. |
1 |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
Ravana 20mg tab. |
2 |
|
MEPACO Company (B.E.) |
Cholenorm tab. |
3 |
|
Jasper Company (B.E.) |
Justechol 20mg. (F. C.T.) |
4 |
|
MEIVO Company (B.E.) |
Ibseverine-s 100mg cab. |
5 |
|
Egyptian group for pharmaceutical Industries (In Vitro) |
Pregabalin 50mg cap. |
6 |
|
Egyptian group for pharmaceutical Industries (In Vitro) |
Pregabalin 100mg cap. |
7 |
|
Egyptian group for pharmaceutical Industries (In Vitro) |
Levetracetam500mg tab. |
8 |
|
Egyptian group for pharmaceutical Industries (In Vitro) |
Meloxicam IS Mg tab. |
9 |
|
Egyptian group for pharmaceutical Industries (In Vitro) |
Health bact400mg tab. |
10 |
|
Grand pharma Company (B.E.) |
Brilliant 100mg. (F. C.T.) |
11 |
|
Grand pharma Company (In Vitro) |
Neuragabalin oral hard gelatin cap. |
12 |
|
شركة القاهره الادوية والصناعات الكيماويه |
Prisoline eye/nasal Drops (sterility test) |
13 |
|
مكتب الاهرام العلمى |
Prisoline eye /nasal Drops (sterility test) |
14 |
2011
|
Company Name |
Trade Name of Product |
No. |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
Olenazapine 10 mg tablets |
1 |
|
Sea Pharma for pharmaceuticals Company(B.E.) |
ELmodoxacin 750 mg tablets |
2 |
|
JASPER Pharmaceutical Industries Company(In Vitro) |
Spazaway tablets |
3 |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
Omeprazole 20mg/ 5ml |
4 |
|
Sea Pharma for pharmaceuticals Company(In Vitro) |
ELmodoxacin 250 mg tablets |
5 |
|
Sea Pharma for pharmaceuticals Company (In Vitro) |
ELmodoxacin 500 mg tablets |
6 |
|
Pharma care ltd. Company (B.E.) |
Montikast 5 mg chewable tablets |
7 |
|
Allam Pharmaceutical Industries Company (In Vitro) |
Uritrospex (Trispium chloride 20mg film coated tablets |
8 |
2012
|
Company Name |
Trade Name of Product |
No. |
|
Egyptian group for pharmaceutical industries Company (In Vitro) |
Alukast tablets |
1 |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
Paroxetine 40mg tablets |
2 |
|
MEDIPHAR LABORATORIES-Lebanon (B.E.) |
ALGODOL (Paracetamol 325 mg + Tramadol HCl 37.5 mg) Capsules |
3 |
|
Chalhoub Pharmaceuticals (CHAPHA) – Lebanon(B.E.) |
Domperidone oral suspension 1mg |
4 |
|
Egyptian group for pharmaceutical industries Company (In Vitro) |
Cold control tablets (Biwaver study) |
5 |
2013
|
Company Name |
Trade Name of Product |
No. |
|
Mepaco company (Bioequivalence study) |
Vedocard tablets |
1 |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
Betahistine Di HCl 24mg |
2 |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
SERNIL tablets |
3 |
2014
|
Company Name |
Trade Name of Product |
No. |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
Proctin tablet |
1 |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
Spiragel tablet |
2 |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
Dislep tablet |
3 |
2015
|
Company Name |
Trade Name of Product |
No. |
|
AUG PHARMA (Microbiological test) |
AUGISPOV Tablets |
1 |
|
AUG PHARMA (Microbiological test) |
AUGISPOV Tablets |
2 |
2017
|
Company Name |
Trade Name of Product |
No. |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
DILO 50 mg Tablets |
1 |
2019
|
Company Name |
Trade Name of Product |
No. |
|
MEDIPHAR LABORATORIES-Lebanon (Bioequivalence study) |
DILO FLEX Tablets (Diclofenac potassium 50 mg + Orphenadrine citrate 50 mg) |
1 |
2022
|
Company Name |
Trade Name of Product |
No. |
|
EL-Nasr for Pharmaceutical Chemicals (Bioequivalence study) |
Lecardopa 275 mg Tablets (Levodopa 250 mg + Carbidopa monohydrate 27 mg eq. to 25 mg Carbidopa) |
 |
 |
 |
|---|---|---|
 |
 |
 |
 |
 |
 |
